Home > Dental Implants
The Surcam® Dental Implant System provides a comprehensive clinical solution for tooth replacement, integrating multiple implant geometries to address a wide range of surgical and restorative indications. Each design is engineered with precision threading, platform compatibility, and controlled surface architecture to ensure predictable mechanical stability and biological integration. Manufactured from Grade 5 titanium (Ti-6Al-4V ELI) under FDA-compliant and ISO 13485-certified conditions, Surcam implants offer proven biocompatibility and long-term durability across all bone types.
Surcam’s PVD TiN Prime Coating Series represents the next step in surface innovation, providing a high-density titanium nitride layer that enhances wear resistance, reduces friction during insertion, and improves torque control. This premium coating supports superior tissue response and aesthetic performance in both surgical and prosthetic components, delivering consistent outcomes in immediate and delayed loading protocols.

Parallel-wall bone-level implant providing optimal load distribution and precise fit in internal-hex workflows.

Self-tapping root-form implant designed for high primary stability in immediate or extraction-site placement.

Cylindrical tissue-level implant engineered for soft-tissue health and predictable one-stage restorations.

Tapered, self-cutting implant with knife-thread design for maximum primary stability in all bone types. SLA surface enhances rapid osseointegration.

Angled implant for maxillary atrophy cases, enabling stable anchorage in the pterygoid plate.

Extra-long implant designed for zygomatic fixation in severe maxillary resorption and full-arch restorations.

Cortical-condensing design offering immediate loading and high stability in soft or low-density bone.

Pterygoid implant with a TiN-coated surface for enhanced hardness, wear resistance, and torque control.

Tissue-level C-Type implant featuring PVD TiN coating for smoother insertion and superior soft-tissue response.

Laser-surface–activated implant with high primary stability and rapid osseointegration. Available in NP 3.5 mm and RP 4.3 mm platforms.

Narrow-platform (NP 3.5 mm) LSA implant providing secure placement in limited ridge width and high primary stability.

Tissue-level C-Type implant with laser-surface activation, RP platform, and optimized soft-tissue contour for long-term integration.

Surcam® implants are manufactured from medical-grade titanium alloy Ti-6Al-4V ELI (Grade 5), recognized for its superior tensile strength, fatigue resistance, and proven biocompatibility. This advanced alloy provides exceptional mechanical stability under functional load while maintaining the corrosion resistance and biological safety required for long-term clinical success.
Higher tensile and fatigue strength than commercially pure titanium ensures structural reliability in all bone types.
CNC-machined under ISO 13485 and FDA-compliant standards to maintain micro-tolerance accuracy across every connection.
Supports immediate and early loading with consistent stability and minimal micro-movement at the bone interface.
Grade 5 titanium exhibits optimal biological response and corrosion resistance, promoting durable bone integration and tissue health.
Validated through ongoing mechanical and clinical testing for predictable outcomes and long-term performance.

Surcam® Prime Series implants and prosthetic components feature a Titanium Nitride (TiN) surface applied through a precision Physical Vapor Deposition (PVD) process. This high-purity, medical-grade coating enhances surface hardness and wear resistance while maintaining full biocompatibility and corrosion protection.
Creates a dense, uniform layer that reduces friction during insertion and prevents micro-wear at the implant–abutment interface.
Significantly increases surface hardness and torque control, extending the mechanical life of implant components under load.
TiN’s inert and hypoallergenic properties support favorable soft-tissue response and long-term biological safety.
The warm gold hue minimizes shine-through under thin gingiva, improving esthetic outcomes in anterior restorations.
Designed to maintain consistent torque values and dimensional accuracy through repeated prosthetic engagement.

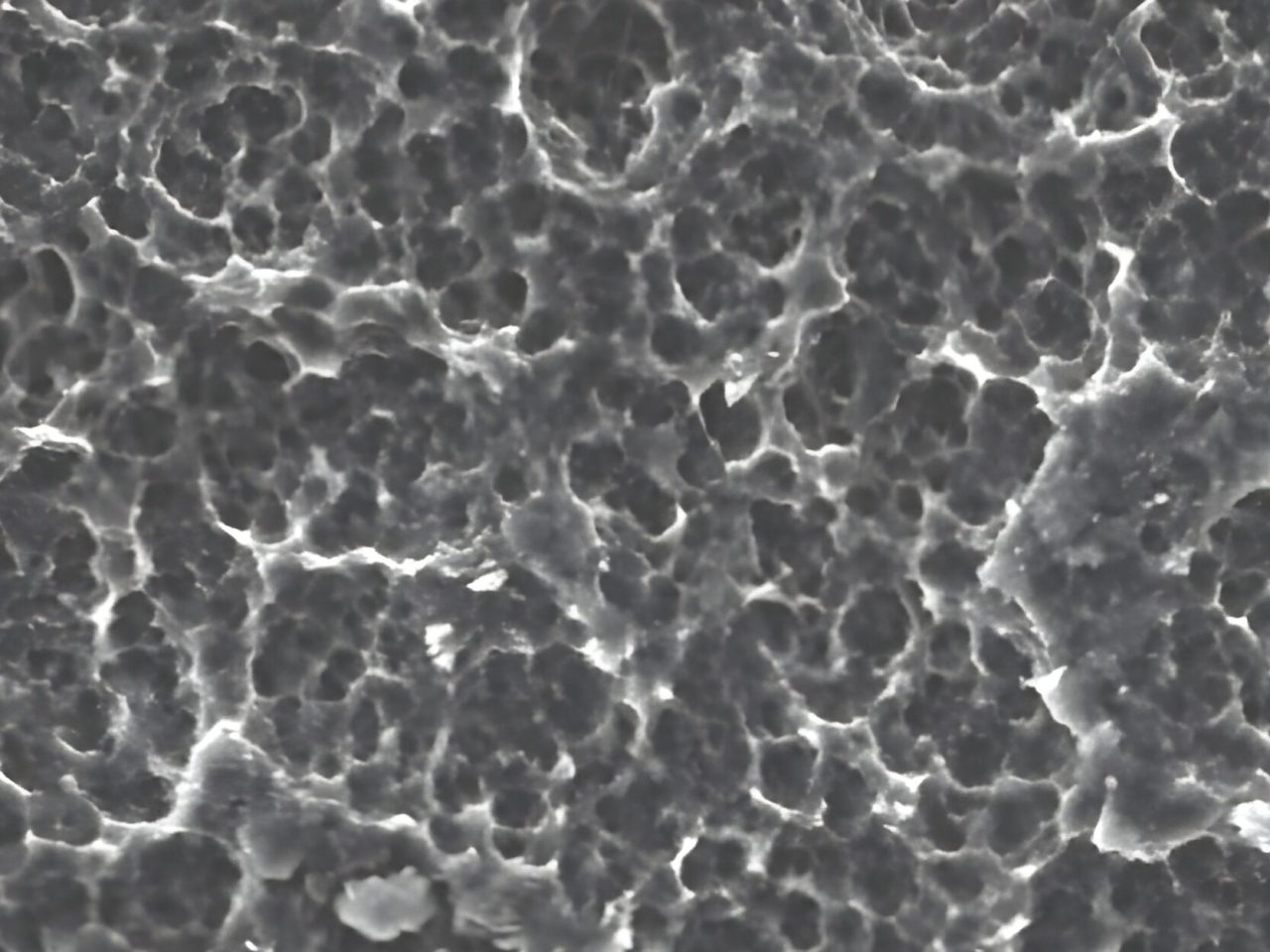
The Surcam® SLA surface is produced through a controlled two-step process involving large-grit sandblasting and acid etching. This technique creates a stable macro- and micro-roughened topography that enhances osteoblast adhesion, early fibrin formation, and bone-to-implant contact. The resulting microstructure supports predictable osseointegration and rapid secondary stability even under early or immediate loading conditions.
Extensive internal validation and clinical feedback confirm high primary stability and consistent long-term integration across all bone types. Surcam® SLA implants demonstrate excellent soft- and hard-tissue response, maintaining crestal bone levels and minimizing biological complications in both single- and multi-unit restorations.
Micro-rough surface enhances cell attachment and bone apposition
Proven early integration and mechanical stability across all bone densities.
Designed for predictable performance in immediate and early loading protocols